What is Bioengineering?
Bioengineering is the discipline that integrates the principles, methods, and design-driven approach of engineering with the knowledge and experimental methodology of the life sciences, with the aim of analyzing, modeling, and designing technological solutions applied to biological systems, assessing not only their functionality, but also their effectiveness and clinical impact.
Biomedical engineering addresses a biological or clinical challenge as a design problem: requirements, constraints, and risks are defined; models are built; architectures and technologies are selected; performance and safety are verified; and real-world impact is validated. In this sense, it reflects the core meaning of engineering itself: the search for solutions to existing problems—transforming needs into systems that work, that are measurable and improvable—through the development and assessment of technologies for prevention, diagnosis, therapy, rehabilitation, and quality of life.
Its defining feature is ongoing collaboration between engineering expertise (electronics, computer science, mechanics, materials, automation) and medical-biological and clinical expertise. However, bioengineering is not simply a “sum of skills”: from the earliest stages of training it promotes a genuinely integrated approach to living systems, viewed as systems.
The GNB promotes and coordinates bioengineering research and training activities carried out in Italy, fostering the growth of a connected and multidisciplinary scientific community.
Examples of technological developments in Bioengineering
Below is an overview (not exhaustive) of the most representative areas, which are often intertwined. While strongly linked to medicine and health, many of these skills also apply to biological systems that are not necessarily human, including veterinary, agri-food, environmental, and bioindustrial domains.
Bioinstrumentation and sensors
Design and validation of sensors, devices, and measurement systems to acquire physiological and biological parameters—from wearables to monitoring systems used in clinical and home settings. This includes measurement reliability, calibration, safety, interoperability, and certification pathways.
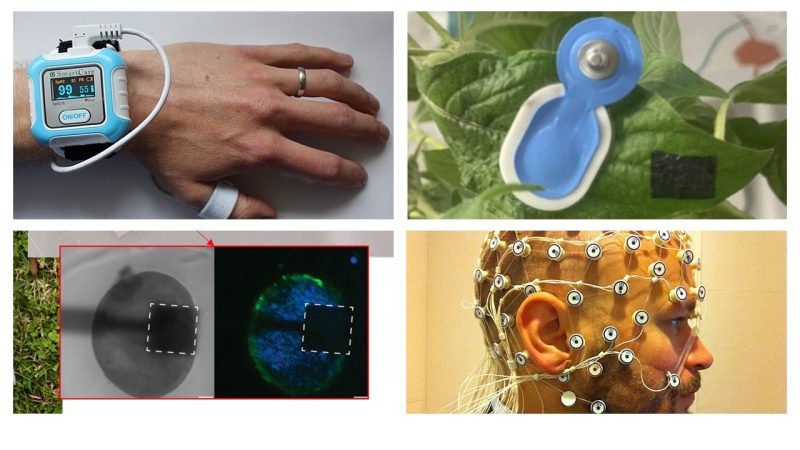
This area is one of the historical pillars of the discipline (often referred to as “bioinstrumentation”) and also finds applications in monitoring non-human biological systems (e.g., food quality, bioprocesses, environment).
Biomedical signal processing and physiological data
Quantitative analysis and interpretation of signals such as ECG/PPG, EEG/MEG, EMG, respiration, pressure, metabolic markers, etc. The goal is to extract biomarkers, estimate physiological/clinical states, support decision-making, and enable adaptive systems (e.g., in rehabilitation or training).
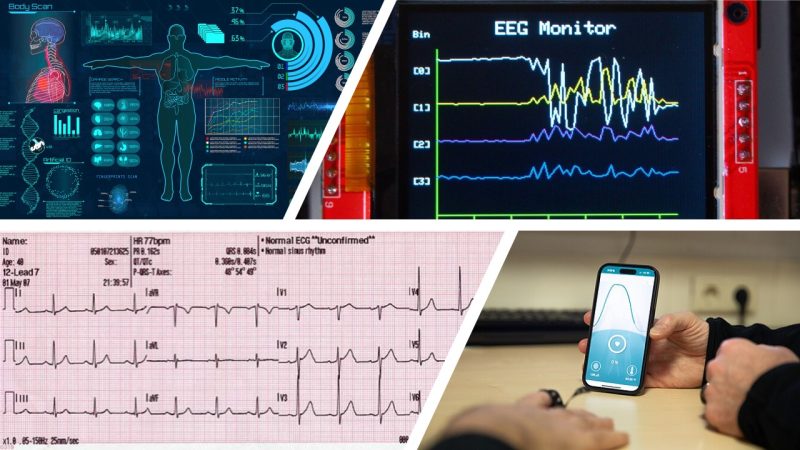
Statistical and machine-learning methods are central, with continued attention to interpretability, bias, and validation.
This area also includes neuroengineering, which integrates measurements, theory, and simulation to understand the computational properties of the nervous system (coding, representation, and information dynamics in neural circuits) and to design neural interfaces and systems.
3) Medical imaging and image computing
Development and optimization of techniques and algorithms for acquiring and processing medical images (MRI, CT, PET, ultrasound, microscopy, multimodal imaging), including reconstruction, registration, segmentation, radiomics, and diagnostic support.
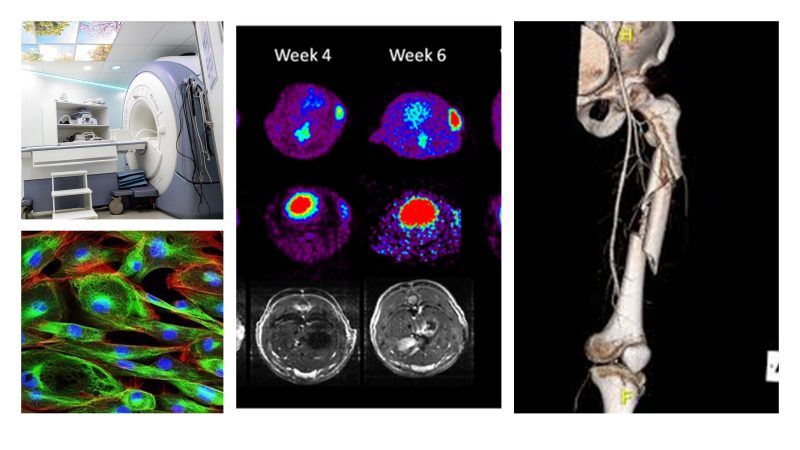
In many modern applications, imaging and AI converge into clinical decision-support tools, with emphasis on data quality, bias, and robustness.
Biomechanics and mechanobiology
Study of biological systems and movement from a mechanical perspective: motion analysis, joint loading, musculoskeletal modeling, tissue dynamics, and physiological fluid dynamics (e.g., hemodynamics).
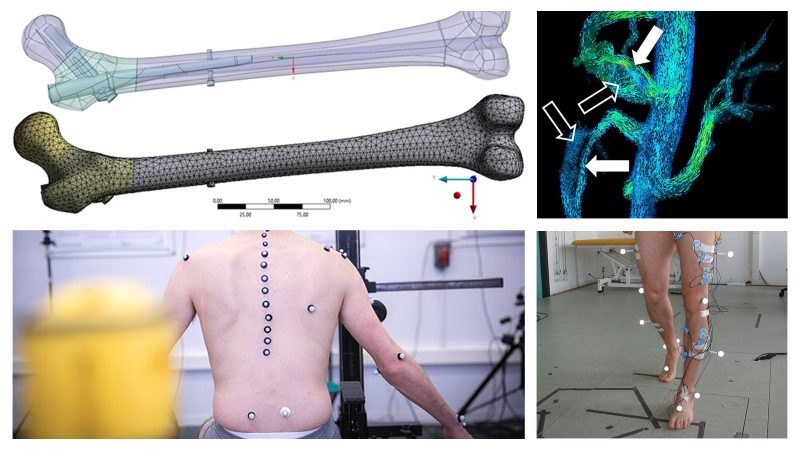
This area is central to orthopedics, sports, ergonomics, injury prevention, and functional assessments.
Biomaterials, prostheses, and implantable devices
Design and characterization of biomaterials and surfaces (biocompatibility, immune response, wear, interaction with cells and tissues), as well as prostheses, stents, valves, scaffolds, and drug-delivery systems.
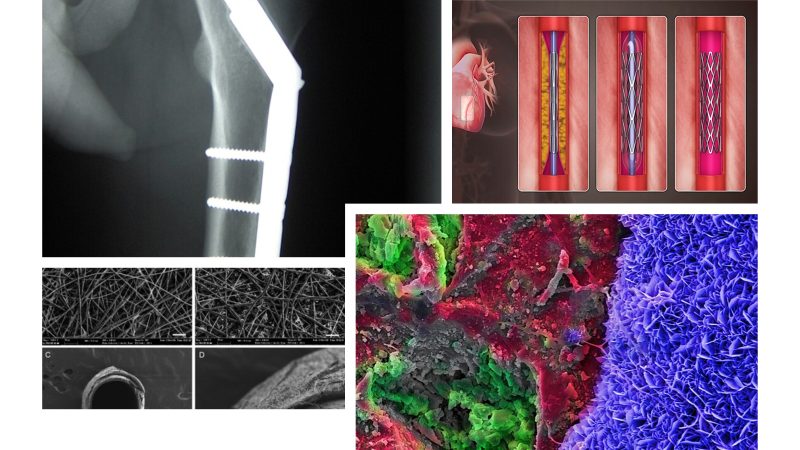
This is a crucial area for clinical and industrial innovation.
Tissue and regenerative engineering
Study of innovative methods to repair or replace tissues using scaffolds, cells, biomaterials, and biochemical/mechanical signals; it includes bioreactors, bioprinting, and regeneration strategies.
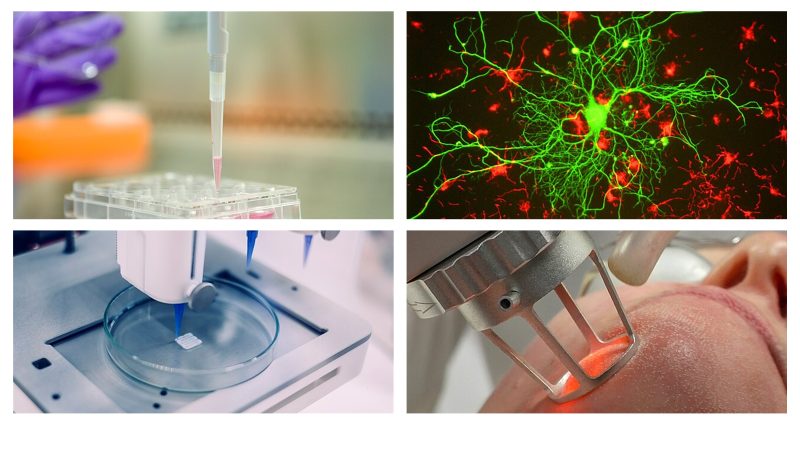
Often associated internationally with “cellular, tissue and genetic engineering,” here engineering design is applied directly to cell biology and physiology.
Bioinformatics, genomics, and computational models for health
Development of computational methods to analyze large-scale biological and clinical data (omics, longitudinal data, electronic health records, real-world data), integrate heterogeneous sources, and build predictive models.

It is a bridge between life sciences, statistics, and AI, with applications ranging from precision medicine to systems biology and the bioeconomy.
Design of technologies for healthcare systems and clinical engineering
Design, evaluation, management, and integration of technologies in healthcare contexts: from defining requirements and constraints (safety, usability, reliability, cybersecurity) to development and performance verification, procurement, maintenance, compliance, and Health Technology Assessment (HTA).
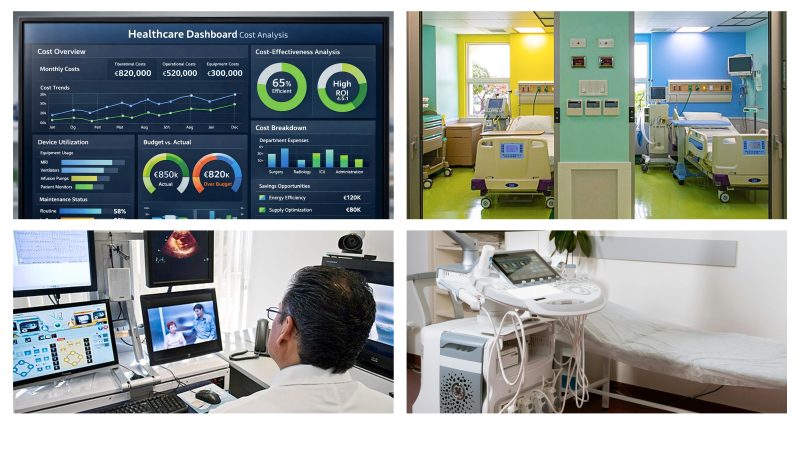
This branch also includes the design and evolution of “standard” medical equipment and electromedical systems used daily in hospitals and in the community (e.g., infusion pumps, oxygen concentrators, dialyzers, and support/monitoring systems), as well as technologies and platforms for in vitro diagnostics (laboratory instrumentation, automation, point-of-care solutions, and measurement systems for tests and analyses). This area—often identified as “clinical engineering”—enables the adoption and real-world implementation of innovations at clinical scale, ensuring continuity between design, safety, and impact in care processes.
Rehabilitation, assistive technology, and medical robotics
Solutions for functional recovery and assistance: intelligent prostheses, exoskeletons, human–machine interfaces, telerehabilitation, and evaluation and training systems. Robotics—whether in the operating room or in rehabilitation—requires particular attention to safety, ergonomics, and clinical outcomes.
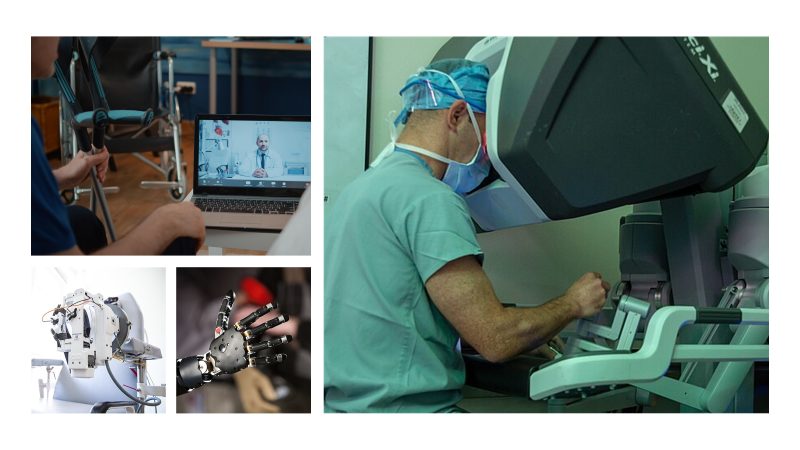
This area also includes bio-inspired and biomimetic robotics, soft robotics, and anthropomorphic/humanoid robotics, where both the robot’s body and control are designed to interact safely and effectively with people and real environments.
Synthetic biology and engineering of living systems (when the “object” is biology itself)
An emerging research direction in which engineering enters biology “from within”: design of genetic circuits, controls, biosystems, and experimental platforms; multiscale modeling and experimental design to understand and reprogram biological functions.

This area strengthens the identity of bioengineering as a discipline of design, not only of application.
A common thread: method, validation, impact
In Italy, bioengineering is expressed through internationally recognized excellence in universities, schools of advanced studies, research institutes, and clinical centers, with particular prominence in bio-inspired and humanoid robotics, neuroengineering, imaging and AI, biomaterials, and health technologies.
Whatever the area, bioengineering shares several key elements:
- design and modeling based on engineering principles (requirements, constraints, trade-offs);
- rigorous experimental and clinical validation (measurements, protocols, reproducibility);
- attention to safety, regulation, ethics, and impact, to transfer solutions toward real-world applications (clinical and industrial).
Within the GNB, this multidisciplinarity translates into community activities, training, and scientific events, with the aim of fostering connections across groups, generations, and research themes.